|
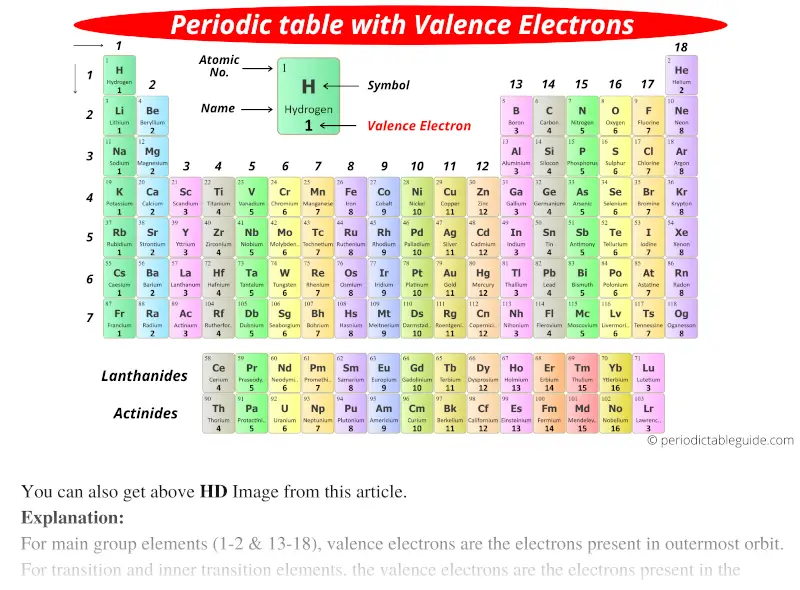
Nevertheless, at GCSE level, it is useful to characterise elements by their valence outermost electrons and electron configuration.įor example, sodium, atomic number 11, has 11 electrons and an electron configuration of 2,8,1. for transition metals, a valence electron can also be in the inner shell, so it can have more than one type of valence). 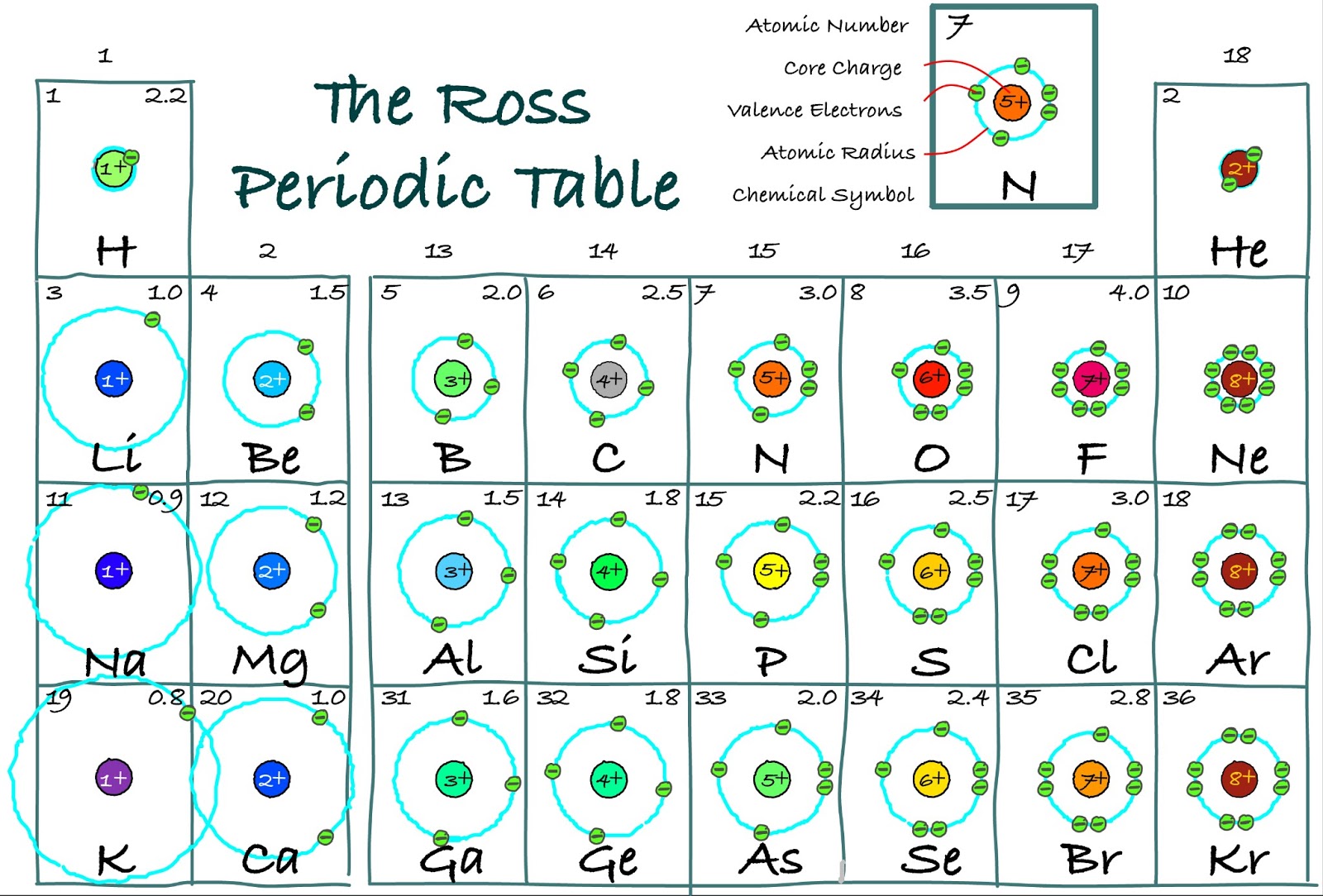
However, there are occasional exceptions to the rule (e.g. In general, t he number of valence electrons corresponds to the Group number. The valence number is the number of electrons that occupy the outermost shell of an atom that can participate in the formation of a chemical bond. 
However, ONLY the electrons that occupy the outermost shell of atoms are involved in electron transfer or electron sharing between atoms.The electrons that orbit closer to the nucleus are, by definition, not in the outermost shell, and are therefore (usually) unaffected during chemical reactions.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
